 For example, if you look at the periodic table, you’ll notice that nickel follows cobalt in the fourth row even though nickel is lighter. Sodium behaved like lithium in its reactions with hydrogen, forming NaH.īut Mendeleev’s most insightful decision was to let properties sometimes trump atomic weight when he placed elements. In contrast, the next element by weight, beryllium (Be), formed the hydride BeH 2.Įach successively heavier element formed different kinds of hydrides until he got to sodium (Na). Mendeleev knew the hydride-a compound of hydrogen with another element-that Li formed had the formula LiH. What sort of chemical properties did Mendeleev have in mind when he developed his table? To get a better idea of the patterns he noticed, let’s start with the metal lithium (Li). But Mendeleev beat Meyer to the punch a year earlier. In Germany, chemist Julius Lothar Meyer was developing his own periodic table that was published in 1870. In 1865, British chemist John Newlands noticed the periodicity of chemical properties and likened the phenomenon to musical octaves, in which the same tone repeats after an increase or decrease of eight notes. By 1829, German chemist Johann Döbereiner had noticed patterns among triplets of elements. As early as 1789, French chemist Antoine Lavoisier had categorized elements into metals, nonmetals, “earths,” and gases, based on their physical and chemical characteristics. And you want to categorize them.īefore Mendeleev came along with his approach, other scientists were attempting to organize the elements. You don’t yet know about protons and therefore atomic numbers, which, for the most part, conveniently run in order from one to 118 from left to right and top to bottom of the table.Īll you know about the elements identified at the time is how they interact with each other, their physical properties, and their relative atomic weights. Imagine for a moment that the distinctive shape of the present-day periodic table with its neat columns and rows doesn’t exist. To understand how Mendeleev created the table in the first place, you have to go back in time and erase what you’ve learned about the table. But who knows, future discoveries could lead to materials that even Mendeleev couldn’t have dreamed of. 
And while the table has been fleshed out over the past century and a half-including the addition of four new elements in 2016-the essence of Mendeleev’s original idea remains. It included the 63 elements that were known at the time, with holes to account for elements that hadn’t yet been discovered. In 1869, Mendeleev published his vision in an early form of the periodic table.  
He found that there was a periodicity to their organization, a repetition of particular chemical properties at regular intervals as atomic weight increased. Time to break out the helium balloons, iron-based sparklers, and calcium-rich ice cream!Īnd whom do we have to thank for the exquisite arrangement of elements? While many scientists contributed to the formation of the table, Russian chemist Dmitri Mendeleev is most often credited for the periodic table’s creation. In celebration of the table, the United Nations proclaimed 2019 as the International Year of the Periodic Table of Chemical Elements. It is 150 years old this year and is holding up well under the test of time-and science. The periodic table has been perhaps as foundational to chemistry as the discovery of DNA has been to biology. You could almost call the table mundane, except really, it’s anything but. 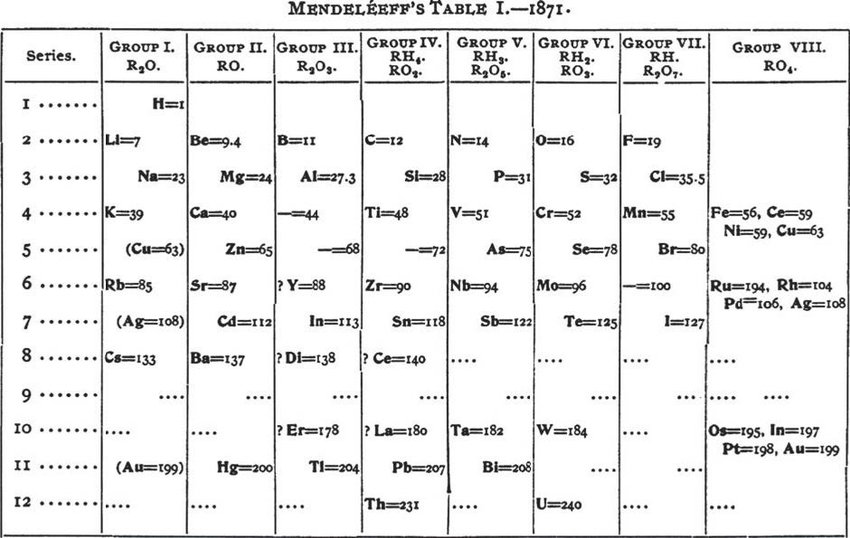
If you google it, you’ll see versions in rainbow colors, or with tiny photos in every box representing each element. You probably have one hanging on your chemistry classroom wall. The periodic table of elements is one of the most recognizable icons of science. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
